For generations, heart disease care followed a one-size-fits-all model. If you had high cholesterol, you received a statin. If your blood pressure was elevated, you got a standard diuretic or beta-blocker. While these approaches saved lives, they left a critical question unanswered: *Why does this medication work wonders for one patient while causing debilitating muscle pain or liver damage in another?*
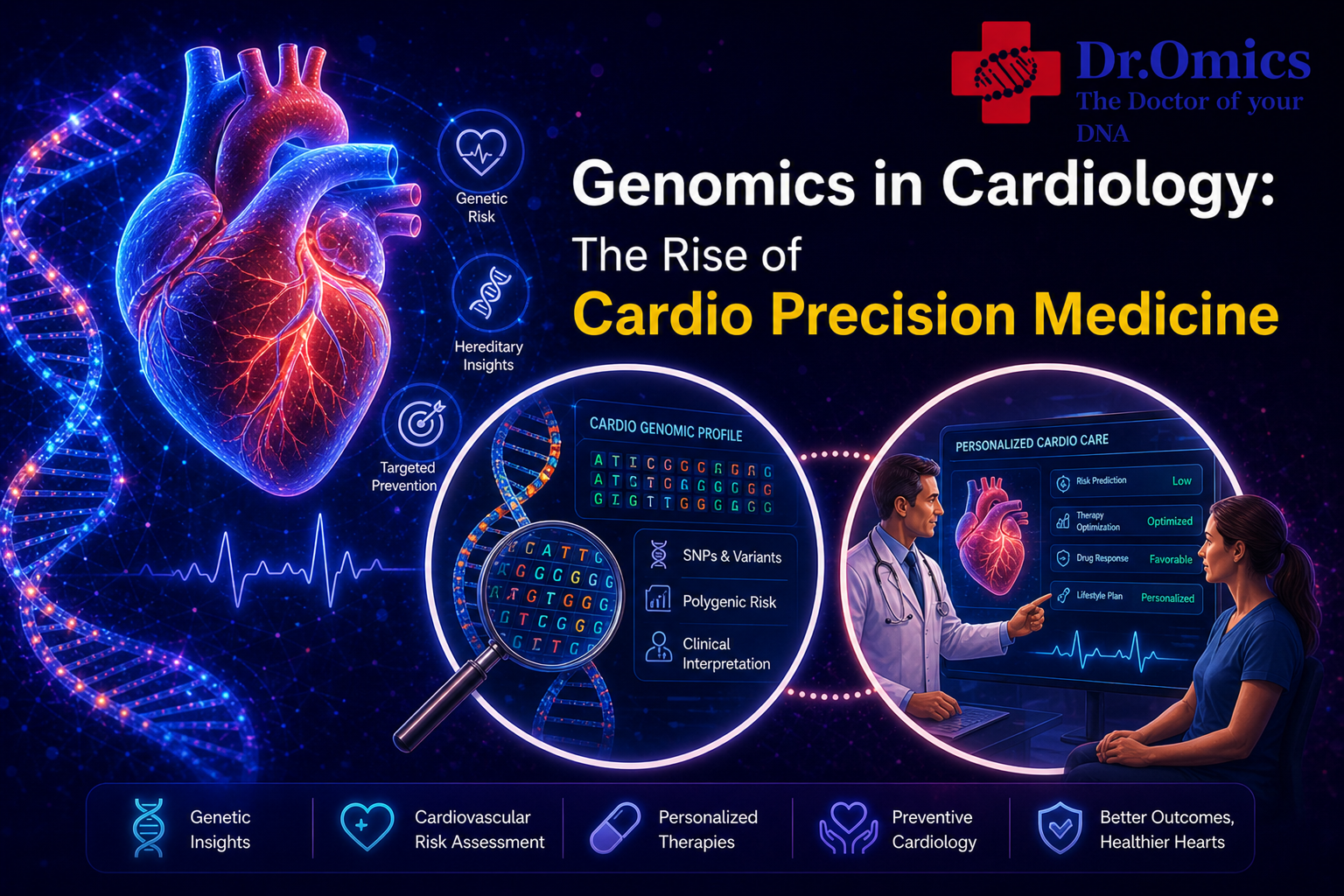
The answer lies deep within our DNA. We are currently witnessing a transformative shift in cardiovascular care, moving away from generalized protocols toward a targeted, individualized approach known as **cardio precision medicine**. By decoding the genetic underpinnings of heart disease, clinicians can now predict risk, select medications, and guide interventions with unprecedented accuracy. At the core of this revolution is **heart genomics**—the study of how your genetic blueprint influences the function of your heart and blood vessels.
Beyond the Stethoscope: The Genetic Heart
The human heart beats roughly 100,000 times per day, driven by a complex electrical system and fueled by metabolic pathways. But subtle variations in your genes can alter these systems dramatically. A single nucleotide change in the *KCNQ1* gene can predispose you to Long QT syndrome, a dangerous arrhythmia. A mutation in *PCSK9* might cause your liver to recycle LDL cholesterol excessively, leading to early heart attacks regardless of diet.
**Heart genomics** identifies these variations. Unlike traditional risk assessments that rely solely on family history and lifestyle factors, a **genetic heart test** provides a direct look at the molecular drivers of your cardiovascular health. It moves the conversation from “You have high cholesterol” to “You have a genetic variant that impairs your ability to clear LDL particles.”
What Is a Genetic Heart Test?
A **genetic heart test** is a specialized laboratory analysis, typically performed on a blood sample or cheek swab, that sequences genes known to influence cardiac structure, rhythm, and metabolism. These panels may look for inherited cardiomyopathies (such as hypertrophic cardiomyopathy), arrhythmia syndromes, or lipid disorders like familial hypercholesterolemia.
But the most actionable area of this field is **cardiovascular pharmacogenomics**—the study of how your genes affect your response to heart medications. This is where a **genetic heart test** moves from abstract risk assessment to immediate clinical utility.
Consider the blood thinner clopidogrel (Plavix). To work, this drug must be activated by an enzyme called CYP2C19, which is produced by the *CYP2C19* gene. Approximately 30% of the population carries a variant that makes this enzyme function poorly. For those individuals, standard clopidogrel is essentially a sugar pill. A **genetic heart test** can identify these “poor metabolizers” before a stent is placed, allowing the cardiologist to choose a different antiplatelet agent like ticagrelor. In the world of **cardio precision medicine**, this distinction can mean the difference between a patent artery and a catastrophic post-stent thrombosis.
Personalized Cardiology in Practice
The goal of **personalized cardiology** is to match every prescription, every dosage, and every lifestyle recommendation to the patient’s unique genomic profile. Beyond clopidogrel, **cardiovascular pharmacogenomics** has delivered several practice-changing insights:
– **Statins (Simvastatin, Atorvastatin):** Variants in the *SLCO1B1* gene dramatically increase the risk of statin-induced myopathy (muscle pain and damage). **Personalized cardiology** protocols now recommend genotyping before prescribing high-dose statins, offering alternatives like pravastatin or rosuvastatin to sensitive patients.
– **Warfarin:** This blood thinner has a narrow therapeutic window. Too little and a clot forms; too much and the patient bleeds. Variants in *VKORC1* and *CYP2C9* determine dosing. **Dna cardiac treatment** guided by **heart genomics** uses an algorithm that incorporates these genetic variants to calculate the precise starting dose, reducing hospitalizations for bleeding or clotting events.
– **Beta-Blockers:** For heart failure patients, variations in the *ADRB1* gene affect how well metoprolol or carvedilol slows heart rate and improves survival. A **genetic heart test** can identify who will benefit most and who may only experience fatigue.
The Rise of DNA Cardiac Treatment
**Dna cardiac treatment** is no longer a futuristic concept; it is a present-day reality for leading academic centers and forward-thinking clinics. For patients with inherited cardiomyopathies, knowing the exact mutation allows for family screening. If a father is found to carry a pathogenic variant for arrhythmogenic right ventricular cardiomyopathy (ARVC), his children can undergo a simple **genetic heart test**. Those who do not carry the mutation can be spared decades of unnecessary tests and anxiety. Those who do can initiate early lifestyle modifications, such as avoiding competitive athletics.
Furthermore, **cardio precision medicine** is revolutionizing the approach to sudden cardiac death. Young athletes who die unexpectedly on the field often have undiagnosed genetic conditions. Post-mortem **heart genomics** can identify the culprit variant, allowing living relatives to be tested and prophylactically treated with implantable defibrillators or medication.
The Future of Cardiovascular Pharmacogenomics
As the cost of sequencing plummets, the routine use of **personalized cardiology** is expanding. We are moving toward a future where a single **genetic heart test** performed in childhood will accompany a patient throughout their life. When that patient turns 60 and needs a blood thinner after a fall, or 50 and requires a statin for borderline cholesterol, the data will already be in the electronic health record.
Clinical trials are currently exploring polygenic risk scores—calculating your cumulative genetic liability for coronary artery disease based on millions of small variants. A high-risk score might motivate a 30-year-old to lower their LDL to 70 mg/dL through aggressive **dna cardiac treatment** with PCSK9 inhibitors or statins, rather than waiting until a heart attack occurs at age 55.
Practical Takeaways
If you have a strong family history of early heart attacks, sudden cardiac death, or high cholesterol resistant to lifestyle changes, ask your provider about **heart genomics**. For patients already on anticoagulation or statins who have experienced side effects or treatment failure, **cardiovascular pharmacogenomics** offers a logical path forward.
However, not every **genetic heart test** is created equal. Seek testing through accredited clinical laboratories, and ensure the interpretation is provided by a cardiologist or genetic counselor trained in **personalized cardiology**. Raw data without context can cause unnecessary anxiety.
The Beat of a New Era
We have spent decades treating the symptoms of heart disease. **Cardio precision medicine** treats the cause. By integrating **heart genomics**, **cardiovascular pharmacogenomics**, and **dna cardiac treatment**, we are finally delivering on the promise of **personalized cardiology**: the right therapy, for the right heart, at the right time.
Your genes do not just tell you where you came from; they tell your cardiologist exactly how to keep you alive. It is time we listened.