For decades, the standard of care for cancer was a three-pronged approach: surgery, chemotherapy, and radiation. While these tools have saved countless lives, they came with a brutal caveat. Chemotherapy, in particular, is a sledgehammer—it kills rapidly dividing cells, but it cannot distinguish between a malignant breast cell and a healthy hair follicle or gut lining. Patients often endured severe toxicity hoping the treatment was working.
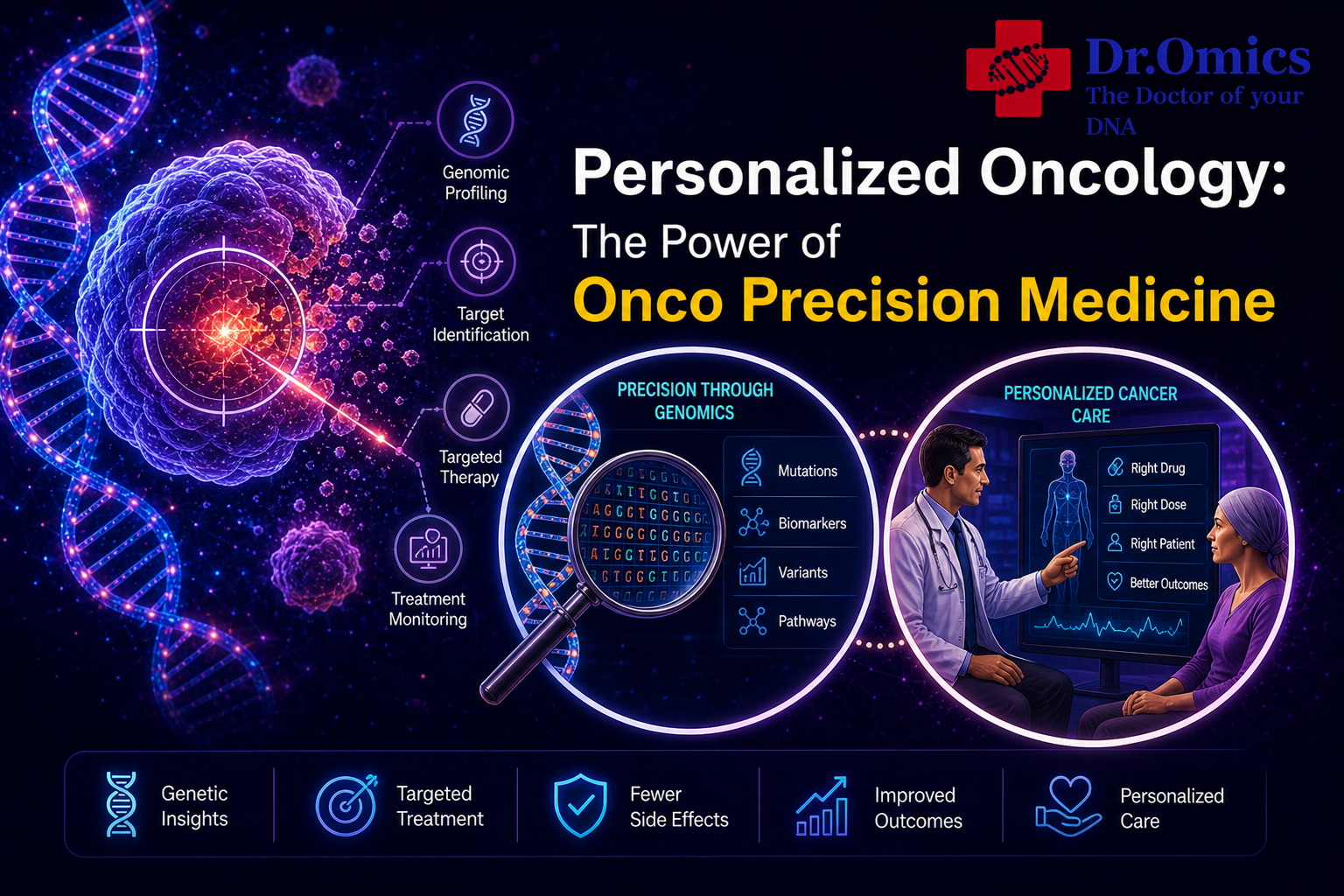
But the landscape of oncology has been rewritten. We have entered the era of **personalized oncology**, a paradigm where treatment is not defined by the location of the tumor, but by its genetic fingerprint. At the heart of this revolution lies **onco precision medicine**—a field that uses the unique DNA of your cancer to select weapons that are targeted, effective, and far less toxic. This is no longer a vision of the future; it is the standard of care for many malignancies today.
The Blueprint of Malignancy
Cancer is, at its core, a disease of the genome. A healthy cell becomes malignant when it accumulates specific mutations in genes that control growth, division, and repair. These mutations might activate an oncogene (like a stuck accelerator pedal) or disable a tumor suppressor gene (like broken brakes).
Traditional chemotherapy attacks all rapidly dividing cells regardless of these mutations. **Onco precision medicine** takes a different approach: it sequences the tumor’s DNA to identify exactly which mutations are driving its growth. This is accomplished through **genomic cancer testing**, a sophisticated laboratory analysis that reads the genetic code of a biopsy sample. Instead of asking “What organ did this cancer come from?” **genomic cancer testing** asks “What genetic errors is this cancer exploiting?”
The Cancer DNA Test: A Molecular Map
A **cancer DNA test** is the foundational tool of **personalized oncology**. Unlike a germline test (which looks at the DNA you inherited from your parents), a **cancer DNA test** typically analyzes the somatic mutations found only within the tumor itself.
These tests come in various sizes. A single-gene test might look for mutations in *EGFR* in non-small cell lung cancer. A small panel might analyze a handful of known drivers like *BRAF* in melanoma or *KRAS* in colorectal cancer. The most advanced approach uses broad panel sequencing—sometimes called comprehensive genomic profiling—which can examine hundreds of cancer-related genes simultaneously, as well as biomarkers like tumor mutational burden (TMB) and microsatellite instability (MSI).
The result is a molecular portrait. That portrait tells the oncologist: “This lung tumor does not rely on the *EGFR* pathway, but it does carry a fusion in the *ROS1* gene.” Or: “This breast cancer is not driven by hormones, but it has a *BRCA1* mutation that makes it vulnerable to a class of drugs called PARP inhibitors.”
Targeted Cancer Therapy: The Intelligent Missile
Once **genomic cancer testing** has identified the driver mutation, the next step is **targeted cancer therapy**. These are drugs designed to interfere with the specific molecular pathways that cancer needs to survive.
Consider the story of imatinib (Gleevec), the original breakthrough in **onco precision medicine**. Chronic myeloid leukemia (CML) is driven almost exclusively by the BCR-ABL fusion gene. Imatinib is a small molecule that fits perfectly into the abnormal ABL kinase active site, blocking it completely. Before imatinib, CML had a five-year survival rate of approximately 30%. Today, patients with CML who receive **targeted cancer therapy** have a near-normal life expectancy and many achieve treatment-free remission.
The same principle applies across tumor types. Lung cancer patients with *EGFR* mutations receive osimertinib. Melanoma patients with *BRAF V600E* mutations receive dabrafenib and trametinib. Colorectal cancer patients with *NTRK* fusions receive larotrectinib. None of these are chemotherapies. They are oral pills or targeted infusions that attack the cancer while largely sparing healthy tissue.
Pharmacogenomics Oncology: Why Dose Matters
Beyond identifying driver mutations, **pharmacogenomics oncology** plays a critical supporting role. This branch of **personalized oncology** examines the patient’s own inherited DNA (germline) to predict how their body will process cancer drugs.
For example, the chemotherapy drug fluorouracil (5-FU) is metabolized by an enzyme called DPYD. Approximately 5-8% of the population carries variants in the *DPYD* gene that reduce enzyme activity. In these patients, a standard dose of 5-FU can lead to severe, life-threatening toxicity including neutropenia, diarrhea, and mucositis. **Pharmacogenomics oncology** guidelines now recommend testing for *DPYD* variants before initiating 5-FU or capecitabine, allowing clinicians to reduce the starting dose by 50-75% in intermediate metabolizers or avoid the drug entirely in poor metabolizers.
Similarly, variations in *UGT1A1* affect the metabolism of irinotecan, a drug used in colorectal and pancreatic cancers. Patients with certain *UGT1A1* variants are at high risk for severe neutropenia and diarrhea. **Pharmacogenomics oncology** allows for dose adjustments upfront, preventing emergency room visits and treatment delays.
Personalized Oncology in Practice
The power of **personalized oncology** extends beyond selecting drugs. It guides surgical decisions, clinical trial enrollment, and even prevention.
For a patient with metastatic lung cancer, receiving a **cancer DNA test** that reveals an *ALK* fusion means they will never receive standard chemotherapy as first-line treatment. Instead, they will receive alectinib or lorlatinib, **targeted cancer therapy** that crosses the blood-brain barrier and controls brain metastases. Their quality of life, progression-free survival, and overall survival are dramatically superior to historical chemotherapy controls.
For a patient with metastatic colorectal cancer, **genomic cancer testing** might reveal that the tumor is microsatellite instability-high (MSI-H). This finding predicts poor response to standard chemotherapy but extraordinary response to immune checkpoint inhibitors (pembrolizumab or nivolumab), which are themselves a form of **targeted cancer therapy** directed at the immune system rather than the tumor directly.
The Future of Onco Precision Medicine
The field is evolving at breakneck speed. Liquid biopsies—**cancer DNA tests** performed on a simple blood draw—now allow oncologists to track how a tumor’s genomic profile changes as it develops resistance to **targeted cancer therapy**. When a patient progresses on osimertinib, a liquid biopsy might reveal a new *EGFR C797S* mutation, guiding the next line of treatment or enrollment in a clinical trial for fourth-generation inhibitors.
Furthermore, **pharmacogenomics oncology** is expanding beyond toxicity prediction. Researchers are exploring how germline variants affect the efficacy of immunotherapy and the risk of immune-related adverse events. The ultimate goal is a fully integrated model: your tumor’s genome selects the drug; your own genome determines the dose.
A Practical Note for Patients
If you or a loved one has received a cancer diagnosis—particularly lung cancer, colorectal cancer, melanoma, breast cancer, or any rare tumor—ask your oncologist about **genomic cancer testing**. In many countries, comprehensive **cancer DNA test** panels are covered by insurance and are often essential for identifying eligibility for **targeted cancer therapy**.
Be aware that turnaround times vary. Standard biopsy-based testing may take one to three weeks. For patients with aggressive disease, rapid panels or liquid biopsies may return results in three to five days.
The Turning Point
We have moved beyond the era of “one-size-fits-all” chemotherapy. **Personalized oncology** and **onco precision medicine** represent a fundamental shift in how we understand and treat cancer. By leveraging **genomic cancer testing**, **cancer DNA test** results, **targeted cancer therapy**, and **pharmacogenomics oncology**, we are finally delivering on a long-held promise: the right treatment, for the right tumor, at the right time.
Cancer is as unique as your fingerprint. Your treatment should be too.